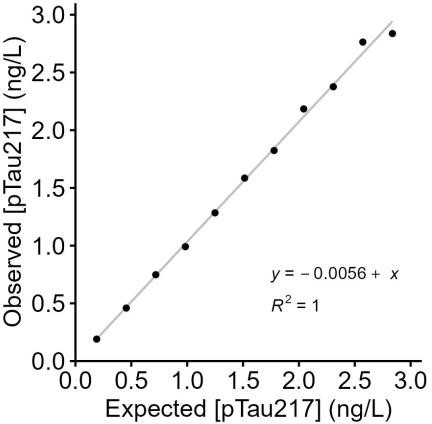
Summary
This report summarizes the clinical and analytical performance of plasma p-Tau217 (ALZpath), including assay accuracy, normal reference range, analytical measurement interval, intra- and inter-laboratory precision, admixture linearity, sample stability, and interference.
Background
Alzheimer's disease (AD) is characterized by extracellular amyloid plaques and intraneuronal neurofibrillary tangles composed of phosphorylated tau (p-Tau) in the brain. Under normal conditions, the tau protein regulates microtubule assembly and helps maintain neuronal axon stability; excessive phosphorylation of tau can contribute to pathology.
In AD, soluble p-Tau accumulates in the cerebrospinal fluid (CSF) and can be measured by lumbar puncture. Sensitive blood-based methods can also measure p-Tau in plasma, and plasma p-Tau217 correlates with amyloid PET imaging as a screening aid for amyloid pathology in cognitively impaired individuals.